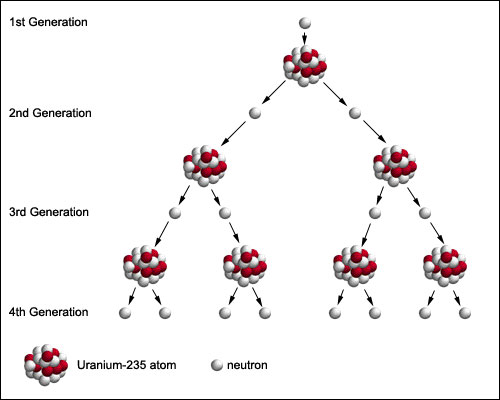
This is why fusion is still in the research and development phase – and fission is already making electricity. The reasons that have made fusion so difficult to achieve to date are the same ones that make it safe: it is a finely balanced reaction which is very sensitive to the conditions – the reaction will die if the plasma is too cold or too hot, or if there is too much fuel or not enough, or too many contaminants, or if the magnetic fields are not set up just right to control the turbulence of the hot plasma. Unlike nuclear fission, the nuclear fusion reaction in a tokamak is an inherently safe reaction. In this fission reaction, about 84 x 10 7 kilojoules of energy is emitted from 1 gram of uranium-235. Splitting of 92 U 235 by absorbing neutron is best example of nuclear fission reaction. As an example, three independent fission yield distributions are shown as a function of mass, A, for neutron-induced fission in Fig. Fusion occurs when two atoms slam together to form a heavier atom, like when two hydrogen atoms fuse to form one helium atom. Example: The reaction of nuclear fission of uranium-235 can be represented as follows. Submarines also need electric power to operate the equipment on board. There is also release of 200MeV energy per fission. Nuclear submarines use nuclear reactors, steam turbines and reduction gearing to drive the main propeller shaft, which provides the forward and reverse thrust in the water (an electric motor drives the same shaft when docking or in an emergency). In conventional nuclear power stations today, there are systems in place to moderate the chain reactions to prevent accident scenarios and stringent security measures to deal with proliferation issues. When 92 U 235 is bombarded with thermal neutron, it splits up into 56 Ba 141 and 36 Kr 92 with the emission of 3 neutrons. This chain reaction is the key to fission reactions, but it can lead to a runaway process resulting in nuclear accidents. The result of the instability is the nucleus breaking up, in any one of many different ways, and producing more neutrons, which in turn hit more uranium atoms and make them unstable and so on. Hydrogen fusion reactions It is one of the nuclear fusion examples. Hence, it is one of the nuclear fusion examples. On average, the hydrogen that converts into helium is about 600 million tons per unit second. Nuclear fusion is a nuclear reaction that combines two or more small atoms to form a large atom. There is a production of an incredibly large amount of heat, one of the forms of energy. The fission process often produce free neutrons and photons. Dorling Kindersley / Getty Images Fission is the. Nuclear fission is a nuclear reaction in which the nucleus of an atom splits into smaller parts. Radioactive decay of both fission products and transuranic elements. Nuclear fission is the splitting of a heavy nucleus into two lighter ones. Nuclear fission is a nuclear reaction that splits a heavy atom into multiple smaller ones. Understand the definition of nuclear fission with examples and an explanation of how the fission process works and why it happens. It is triggered by uranium absorbing a neutron, which renders the nucleus unstable. Nuclear reactors work by containing and controlling the physical process of nuclear fission. 
Fission and chain reactionsįission is the nuclear process that is currently run in nuclear power plants. Both reactions release energy which, in a power plant, would be used to boil water to drive a steam generator, thus producing electricity. However, fusion is combining light atoms, for example two hydrogen isotopes, deuterium and tritium, to form the heavier helium. The nucleus of an uranium atom splits into smaller isotopes krypton and barium, producing free neutrons and. \): The fission of a large nucleus, such as U-235, produces two or three neutrons, each of which is capable of causing fission of another nucleus by the reactions shown.In fission, energy is gained by splitting heavy atoms, for example uranium, into smaller atoms such as iodine, caesium, strontium, xenon and barium, to name just a few. Illustration of a radioactive decay process.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
